Key points
- Tuberculosis Trials Consortium (TBTC) is a unique collaboration of researchers from CDC, domestic and international public health departments and academic medical centers, and selected Veterans Administration medical centers.
- TBTC’s mission is to conduct programmatically relevant research concerning the diagnosis, clinical management, and prevention of tuberculosis (TB) infection and disease.
- The current research cycle began in January 2021 and will end in December 2030.

Overview
CDC assesses the need for and conducts studies of new or existing drugs and regimens used in the prevention and treatment of TB, including:
- Dosage,
- Duration,
- Pharmacokinetics, and
- Safety.
Currently, most of these trials and studies are conducted by TBTC.
Misson
TBTC’s mission is to conduct programmatically relevant research concerning the diagnosis, clinical management, and prevention of tuberculosis infection and disease.
The work of TBTC:
- Expands clinical and epidemiologic knowledge of TB,
- Integrates research into the care of persons with TB infection and disease,
- Promotes research within local TB programs through collaboration on clinical research of relevance to public health settings, and
- Provides a forum for international collaborative research of importance to both domestic and international TB control.
Roles
CDC
The CDC oversees, and collaborates in, the work of TBTC. The TBTC CDC team is composed of medical officers, epidemiologists, trialists, health scientists, microbiologists, data analysts, data managers, programmers, data clerks, multiple public health students, administrative support staff, and others. The CDC team supports the TBTC by:
- Functioning as the TBTC's Data and Coordinating Center;
- Participating in protocol development;
- Guiding implementation, analysis, and interpretation of each trial;
- Conducting the central monitoring for all TBTC trials;
- Performing data management;
- Managing drug supplies;
- Establishing site laboratory standards;
- Executing pharmacovigilance for TBTC trials;
- Presenting scientific data at conferences and in author publications; and
- Coordinating administrative, regulatory, and fiscal support for the TBTC.
Committees
Several working committees make up the governance of TBTC:
- Steering Committee: made of representatives from all those engaged, makes major decisions for the group
- Executive Affairs Group: serves as the executive arm of the Steering Committee and conducts the Consortium's day-to-day administrative business
- Core Science Group: develops the scientific program of research
- Implementation and Quality Committee: supervises the conduct and quality of ongoing studies
- Publications and Presentations Committee: assures quality and equity in TBTC's reporting of its work
- Advocacy and External Relations Committee: represents the TBTC to outside entities and supports the Consortium's community engagement activities
CDC staff are represented and participate on all committees.
Other Partners
Developing new TB treatment and prevention strategies depends upon collaboration among academics, private sector and government researchers, public health departments, manufacturers of pharmaceuticals, regulatory agencies, and non-governmental organizations. CDC works closely with such organizations as:
- U.S. Food and Drug Administration,
- National Institutes of Health and its National Institute of Allergy and Infectious Diseases,
- Global Alliance for TB Drug Development, and
- Other partners within and outside the United States.
Such partnerships build upon the long tradition of collaboration in pursuit of important public health goals.
Research priorities
The current pipeline of new and re-purposed anti-TB drug candidates is the most promising in 40 years. Advances in TB clinical trials science have fostered the progress of these agents, supported by many members of the global TB control community.
With commitment and support from CDC, the TBTC adds importantly to the resources available for these clinical trials and is expected to continue to make useful contributions to TB treatment, prevention, and control.
Current activities include:
CRUSH-TB/Study 38
- CRUSH-TB (Combination Regimens for Shortening Tuberculosis Treatment)/Study 38 is a phase 2C trial aims to assess the efficacy and safety of several regimens based on novel agents in the treatment of drug-sensitive TB disease.
- The trial compares the effectiveness and safety of new 4-month bedaquiline, moxifloxacin, and pyrazinamide-based regimens to the standard of care 6-month regimen for TB disease.
- ClinicalTrials.gov Identifier: NCT05766267
ASTERoiD/Study 37
- ASTERoidD (Assessment of the Safety, Tolerability, and Effectiveness of Rifapentine Given Daily for Latent TB Infection)/TBTC Study 37 is an open label, multi-center, phase 3 randomized controlled non-inferiority trial that compares the safety and effectiveness of a 6-week regimen of daily rifapentine against the current standard of 12-16 weeks of rifamycin-based treatment for latent TB infection.
- ClinicalTrials.gov Identifier: NCT03474029
TBTC Study 35
- TBTC Study 35 is a Phase I/2 open-label, single arm, exposure-controlled study to determine appropriate dosing of a novel water-dispersible, child-friendly formulation of rifapentine with isoniazid in children aged 0-12 years.
- The trial aims also to assess safety of this formulation in HIV-infected and HIV-uninfected children.
- If successful, the trial will contribute to global availability of a pediatric formulation that can be used to treat latent TB infection in young children.
- ClinicalTrials.gov Identifier: NCT03730181
TBTC Study 31
- TBTC Study 31 was a large trial of a 4-month rifapentine-based regimen for treatment of drug-sensitive TB, conducted in collaboration with the National Institute of Allergy and Infectious Diseases, AIDS Clinical Trials Group (as ACTG A5349), demonstrated non-inferiority of one of two test regimens.
- The trial has concluded, and many analytic activities are currently underway.
- ClinicalTrials.gov Identifier: NCT02410772
Timeline
TBTC 1993-1998
In 1993-94, CDC announced a competitive solicitation to fund a group of TB researchers and sites capable of conducting TB clinical trials; successful applicants were funded for a period of 5 years. CDC selected sites that:
- Provided access to significant numbers of TB patients,
- Had experience with clinical trials,
- Demonstrated qualifications of the trial team, and
- Submitted robust plans for recruitment, management, and follow-up of patients.
U.S. Public Health Service Study 22
- Planning together, the researchers and CDC decided to conduct first a randomized, clinical trial comparing a once-weekly, rifapentine-based, continuation-phase regimen to the then-standard, twice-weekly, rifampin-based "Denver" regimen, both given for the last 4 months of the 6-month treatment for TB.
- This trial, USPHS Study 22, began in 1995, enrolled 1,075 TB patients from the United States and Canada, and reported final results in 2002.
Creation of TBTC
In 1997-98, CDC and the Study 22 investigators reorganized their collaboration as a structured consortium, creating the TBTC with the adoption of formal by-laws in 1998. The bylaws were updated in 2012 and again in 2021.
TBTC 1999-2009
- In 1999, the TBTC underwent its second competitive process to select the clinical trial sites that would comprise the TBTC during 1999–2009.
- The selection of sites at that time resulted in a consortium of 23 centers located in the United States and Canada.
- In 2003, the consortium gained an international presence by adding sites in Brazil, Spain, and Sub-Saharan Africa.
TBTC 2010-2020
- In 2009, the TBTC underwent its third re-competition.
- During 2010–2020, TBTC’s international presence expanded from a few clinical study sites located outside of North America, to sites in Peru, Spain, South Africa (two sites), Uganda, Kenya, Vietnam, and China (Hong Kong).
- The TBTC 2010–2020 also included U.S. sites in New York, Washington DC, Texas (four sites), Colorado, and Tennessee.
TBTC 2021-2030
- In 2021, CDC announced the sites for the research cycle through December 2030.
Accomplishments
Since its inception, TBTC clinical trials have had significant impact on TB treatment and prevention:
- U.S. Public Health Service Study 22 substantially influenced American Thoracic Society (ATS)/CDC/Infectious Disease Society of America (IDSA) guidelines for treatment of TB disease.
- TBTC Study 23 results have led to modification of CDC's recommendations for treatment of TB and HIV.
- TBTC Studies 26 and 33 led to updates in National Tuberculosis Controllers Association/CDC guidelines for treatment of latent TB infection.
- TBTC Study 31/ACTG A5349 was the first trial to achieve success with a 4-month treatment regimen for active TB; it demonstrated that a four-month daily treatment regimen containing a combination of high-dose rifapentine and moxifloxacin is non-inferior to the standard six-month daily regimen for drug-susceptible TB disease. The results of this study led to CDC's publication of interim guidance for this 4-month regimen and updated WHO treatment guidelines.
- TBTC Study 34 influenced the 2017 ATS/CDC/IDSA guidelines for TB diagnosis.
Future direction
TBTC clinical trials have enrolled more than 16,000 patients and volunteers over the past 20 years. CDC is confident that TBTC will continue to contribute to development of stronger approaches to treatment and prevention of TB over the next decade.
How it's funded
TBTC sites are funded in one of two ways, either through
- Individual contracts with CDC or as
- Part of a sub-network coordinated by investigators at the Washington, D.C. Veterans Affairs (VA) Medical Center.
Every 10 years, both sides of the consortium undergo re-competition. The current cycle began January 2021 and will end in December 2030.
Funding recipients
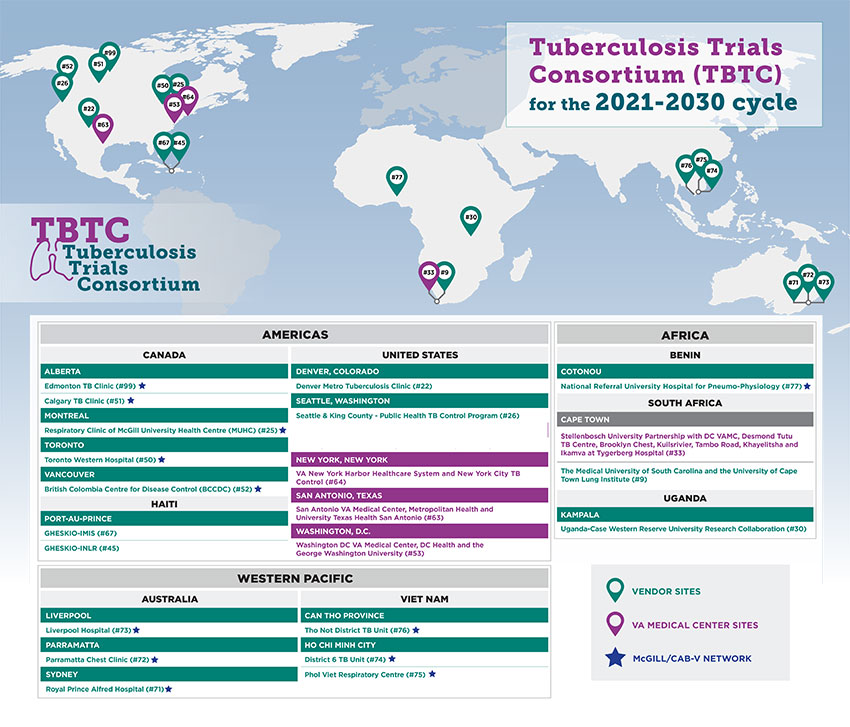
2021-2030 TBTC sites are located in Australia, Benin, Canada, Haiti, South Africa, Uganda, the United States, and Vietnam.
Case-Kampala
- Case Western Reserve University and the Uganda-Case Western Reserve University Research Collaboration, in Kampala, Uganda (Site #30)
- Principal Investigators (PI): John L. Johnson, MD; Harriet Mayanja-Kizza; Moses L. Joloba
Cornell/Haiti
- Joan & Sanford I. Weill Medical College of Cornell University (Site #45)
- PI: Daniel Fitzgerald, MD
- GHESKIO Centers in Port-au-Prince, Haiti (Site #67)
- PI: Jean William Pape, MD
Denver
- The Denver Metro Tuberculosis Clinic, part of Denver Public Health, in Denver, Colorado (Site #22),
- PI: Robert Belknap, MD
McGill/CAB-V
- The CAB-V Network is an established group of investigators at 8 sites in the 5 largest cities in Canada, and the largest cities in Australia, Benin, and Vietnam.
- PIs: Dick Menzies (McGill University); James Johnston (University of British Columbia) and Greg Fox (University of Sydney), including the following:
Canadian sites
- McGill University Health Centre tertiary hospital, Montreal, Canada (Site #25)
- PI: Dick Menzies, MD
- Edmonton TB Clinic, Alberta, Canada (Site #99)
- PI: Richard Long, MD
- Toronto Western Hospital, Toronto, Canada (Site #50)
- PI: Sarah Brode, MD
- Calgary TB Clinic Alberta, Canada (Site #51)
- PI: Dina Fisher, MD
- British Columbia Centre for Disease Control, Vancouver, Canada (Site #52)
- PI: James Johnston, MD
Australian sites
- Royal Prince Alfred Hospital, Sydney, Australia (Site #71)
- PI: Greg Fox, MD
- Parramatta Chest Clinic Parramatta, Australia (Site #72)
- PI: Jin-Gun Cho, MD
- Liverpool Hospital, Liverpool, Australia (Site #73)
- PI: Harrington Zinta, MD
Vietnam Sites
- District 6 TB Unit, Ho Chi Minh City, Vietnam (Site #74)
- PI: Greg Fox, MD
- Phoi Viet Respiratory Centre, Ho Chi Minh City, Vietnam (Site #75)
- PI: Greg Fox, MD
- Thot Not District TB Unit, Can Tho Province (Site #76)
- PI: Greg Fox, MD
Benin Site
- National Referral University Hospital for Pneumo-physiology, Cotonou, Benin (Site #77)
- PI: Menonli Adjobimey, MD
MUSC/UCTLI
- The Medical University of South Carolina (MUSC)
- PI: Susan Dorman, MD
- University of Cape Town Lung Institute (UCTLI) in Cape Town, South Africa (Site #09)
- PI: Dr. Rodney Dawson (Bio Analytical Research Corporation partnership)
Seattle
- The Seattle & King County – Public Health TB Control Program, in Seattle, Washington (Site #26),
- PIs: Masa Narita, MD and David Horne, MD
Veterans Affairs (VA) Medical Center sites
- San Antonio: San Antonio VA Medical Center, University of Texas/San Antonio VA Hospital (Site #63),
- PI: Jose Cadena Zuluaga, MD
- Stellenbosch: Stellenbosch University, Cape Town, South Africa (administered via sub-contract under the Washington DC VA Medical Center)
- PI: Anneke Hesseling, MD
- PI: Anneke Hesseling, MD
- Washington DC: Washington DC VA Medical Center, Washington, District of Columbia, coordinating center for the VA Affiliates Group and George Washington University Medical Center (Site# 53)
- PIs: Debra Benator, MD (Washington DC VA Medical Center) and Afsoon Robers, MD (George Washington University Medical Center)
- New York: VA New York Harbor Healthcare System (Site #64), in collaboration with New York City Bureau of Tuberculosis Control
- PIs: Benjamin Wu, MD (New York Harbor Healthcare System) and Joseph Burzynski, MD (New York City Bureau of Tuberculosis Control)
Map of TBTC sites for the 2021-2030 cycle