Purpose
The goal of the CDC Vitamin D Standardization and Certification Program (VDSCP) is to improve the analytical accuracy and reliability of clinical vitamin D tests.

CDC Vitamin D Standardization - Certification Program (VDSCP)
Standardization of Measurement Procedures
CDC help laboratories and assay manufacturers with their calibration and analytical performance. The VDSCP assesses the assay performance (bias and imprecision) throughout one year (initially for four consecutive quarters).
Participants in VDSCP obtain detailed information about the analytical accuracy and precision of their vitamin D tests. Tests that meet criteria for analytical accuracy and precision are certified.
VDSCP has several key characteristics that are not available with other programs:
- It provides a panel of single-donor sera covering a wide range of vitamin D concentrations. This gives detailed information about measurement performance at multiple concentration levels and helps identify potential sources of measurement bias.
- It assesses measurement accuracy and imprecision by collecting data from replicate measurements.
- It monitors the consistency of participants' vitamin D measurements by assessing measurement performance quarterly.
Assessments of analytical bias and precision are performed using well-recognized and established protocols.
The typical procedure to achieve VDSCP standardization certification starts with an initial assessment of the participant's testing performance. That is followed by enrollment in the actual certification process. Participants may, however, choose to do only one or the other.
Spotlight
How this is done:
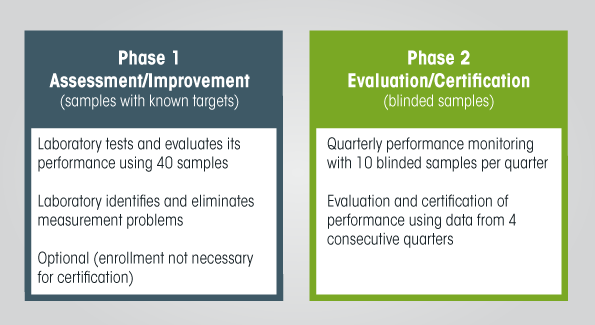
In the initial assessment process - Assessment and Improvement of Analytical Performance (Phase 1):
- participants submit a request to VDSCP for a set of samples to measure;
- participants measure these samples and compare their results against those assigned by the CDC reference method to uncover analytical performance improvements that might be needed.
VDSCP suggests following Clinical and Laboratory Standards Institute (CLSI) Protocol EP9-A, "Method comparison and bias estimation using patient samples," for performing initial assessments. This document recommends a minimum of 40 samples. VDSCP can provide participants with any numbers of different samples, up to 120 per set.
In the actual certification process - Verification and Certification of Analytical Performance (Phase 2:
- CDC VDSCP sends 10 blinded serum samples to participants every quarter.
- Participants measure these samples and report the results to VDSCP.
- Each participant then receives a report about their measurement accuracy and imprecision every quarter.
VDSCP uses results from four consecutive quarters to assess measurement accuracy and imprecision, then issues a comprehensive report for each participant. Participants meeting analytical performance criteria for bias and imprecision are certified.
Criteria used to certify Vitamin D
As part of the certification process, the accuracy of tests for total 25-hydroxyvitamin D are compared with predefined analytical performance criteria. Tests meeting these criteria are considered standardized by VDSCP.
| Maximum Allowable Bias* | Maximum Allowable Imprecision (Coefficient of Variation) |
|---|---|
| ≤5.0% | <10.0% |
* Bias to the reference method.
Validation/certification protocols
The CDC VDSCP program for total 25-hydroxyvitamin D (25OHD) started in December 2012.
- Participant Protocol for Vitamin D Standardization-Certification Program – Updated April 2023
- VDSCP Vitamin D Certified Assays – Updated November 2023
